 :max_bytes(150000):strip_icc()/atomic-structure-conceptual-artwork-99312661-58af58c75f9b5860467ff472.jpg)
Starting with Rutherford's work, Chadwick continued experimenting to determine the excess mass in the nucleus that wasn't accounted for by protons. 
James Chadwick won a Nobel Prize in 1935 for his discovery of the neutron. While the Bohr model holds true for hydrogen, it isn't accurate for multi-electron elements. When an electron changed energy levels, it gained or lost energy, which could be emitted in the form of light. In support of his theory, Bohr examined the behavior of electrons in hydrogen molecules and noted that they followed distinct energy levels. Bohr, who was also one of Rutherford's students, suggested that electrostatic forces kept electrons in circular orbit around the positively charged central nucleus. He defined an atom as the smallest indivisible particle. Though some of his conclusions were incorrect, his contributions were vital. Niels Bohr: Introducing Quantum Physics The Bohr model for a Hydrogen atom with its electron moving between energy levelsĪs quantum mechanics progressed through the work of Albert Einstein and Max Planck, Niels Bohr updated the atomic model in 1913 to account for quantum phenomena. Dalton's atomic model sets up the building blocks for others to improve on. 
He proposed a planetary model of atoms where a cloud of electrons surrounded a central, positively charged nucleus. This deflection could only occur if the positive components of atoms were located in a small area, which Rutherford assumed to be the center. He shot small, positively charged alpha particles at thin sheets of gold foil and noted that some particles were sharply deflected. In 1909, one of Thomson's students, Ernest Rutherford, determined that the positive charge of atoms was located in a central nucleus. (He likened the relationship of electrons to the sea of positive charge to that of plums in plum pudding.)Įrnest Rutherford: Gold Foil Experiment and the Nucleus Thus, Thomson developed the "plum-pudding" model of negatively charged electrons floating in a sea of positive charge. Thomson knew that atoms had a net neutral charge, but he only knew that negative particles existed. By the late 1800’s, John Dalton’s view of atoms as the smallest particles that made up all matter had held sway for about 100 years, but that idea was about to be challenged. Thomson discovered the electron in 1897, and was the first to learn that atoms weren't actually "uncuttable" as initially thought. This module is an updated version of Atomic Theory I. Thomson: The Plum-Pudding Model and Electrons Tomson's Plum-Pudding Model As early as 1905, Albert Einstein used Brownian Motion to predict the size of atoms and molecules. 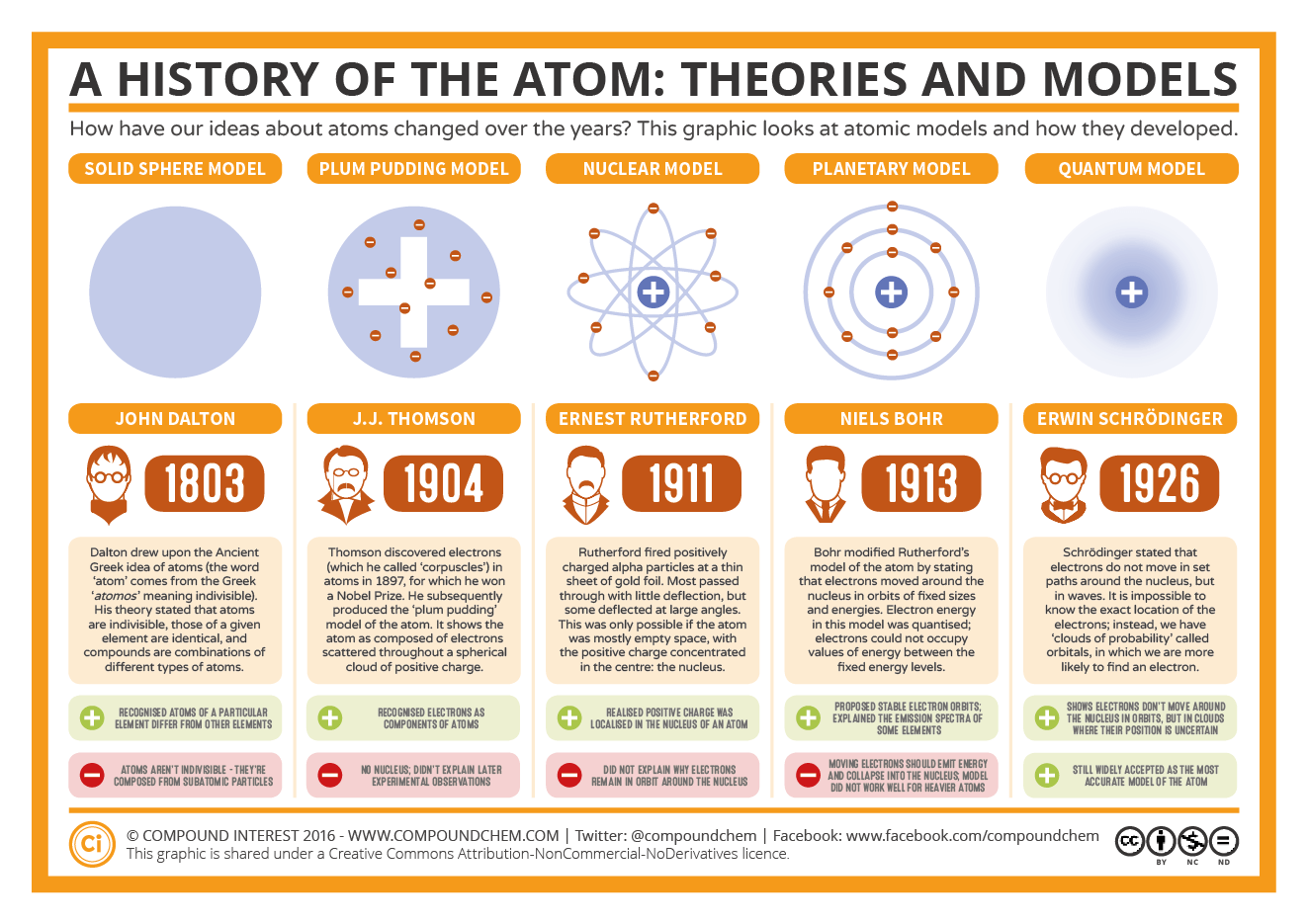
The particles followed complex paths, dubbed Brownian Motion. This allowed Avogadro to take more accurate atomic measurements of gases than Dalton, and differentiate atoms from molecules.Ī Scottish botanist, Robert Brown, studied the motion of tiny pollen particles in water in 1827. In 1811, Amedeo Avogadro studied gases and determined that the amount of volume a gas occupies is not determined by the mass of the gas. He shared the 1933 Nobel Prize for Physics with British physicist P.A.M. Dalton was unable to distinguish between atoms and molecules (groups of atoms). Erwin Schrödinger (born August 12, 1887, Vienna, Austriadied January 4, 1961, Vienna) Austrian theoretical physicist who contributed to the wave theory of matter and to other fundamentals of quantum mechanics. For example, it was Charles-Augustin de Coulomb who first determined the mathematical equation which could accurately describe the electrostatic potential between charged particles that make up an atom: Such fundamental formulas are powerful tools for helping scientists understand the interactions within and processes of both microscopic and macroscopic systems. The goal of a mathematical model is to express the underlying rules that govern atomic processes in a formula that can then predict and describe the behavior of atoms (such as is seen with the list of Bohr's mathematical models for the atom). Mathematical models of atomic processes have become more thorough and sophisticated as knowledge of atomic properties has grown.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
